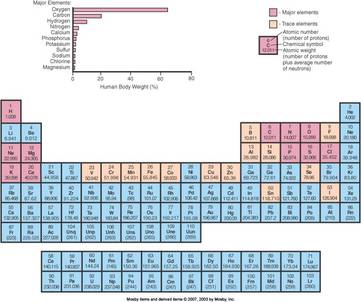
Element
–“Pure”
–Cannot be broken down into two or more different substances
–26 elements in the human body
–11 “major elements,” four of which make up 96% of the human body
–15 “trace elements” that make up less than 2% of body weight
Compound
–Atoms of two or more elements joined to form chemical combinations
Atoms
–The concept of an atom was proposed by the English chemist John Dalton
Atomic structure
–Atoms contain several different kinds of subatomic particles; the most important are the following:
Protons (+ or p)
–positively charged subatomic particles found in the nucleus
Neutrons (n)
–neutral subatomic particles found in the nucleus
Electrons (– or e)
–negatively charged subatomic particles found in the electron cloud
Atomic number and atomic weight
–Atomic number
The number of protons in an atom’s nucleus
The atomic number is critically important; it identifies the kind of element
–Atomic weight
The mass of a single atom
It is equal to the number of protons plus the number of neutrons in the nucleus (p + n)
–“Pure”
–Cannot be broken down into two or more different substances
–26 elements in the human body
–11 “major elements,” four of which make up 96% of the human body
–15 “trace elements” that make up less than 2% of body weight
Compound
–Atoms of two or more elements joined to form chemical combinations
Atoms
–The concept of an atom was proposed by the English chemist John Dalton
Atomic structure
–Atoms contain several different kinds of subatomic particles; the most important are the following:
Protons (+ or p)
–positively charged subatomic particles found in the nucleus
Neutrons (n)
–neutral subatomic particles found in the nucleus
Electrons (– or e)
–negatively charged subatomic particles found in the electron cloud
Atomic number and atomic weight
–Atomic number
The number of protons in an atom’s nucleus
The atomic number is critically important; it identifies the kind of element
–Atomic weight
The mass of a single atom
It is equal to the number of protons plus the number of neutrons in the nucleus (p + n)
Energy levels
–The total number of electrons in an atom equals the number of protons in the nucleus
–The electrons form a “cloud” around the nucleus
Chemical bonds—two types unite atoms into molecules:
–Ionic, or electrovalent, bond
Formed by transfer of electrons; strong electrostatic force that binds positively and negatively charged ions together
–Covalent bond
Formed by sharing of electron pairs between atoms
–The total number of electrons in an atom equals the number of protons in the nucleus
–The electrons form a “cloud” around the nucleus
Chemical bonds—two types unite atoms into molecules:
–Ionic, or electrovalent, bond
Formed by transfer of electrons; strong electrostatic force that binds positively and negatively charged ions together
–Covalent bond
Formed by sharing of electron pairs between atoms
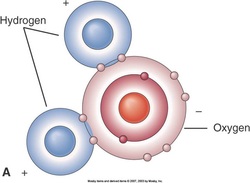
Hydrogen bond
–Much weaker than ionic or covalent bonds
–Results from unequal charge distribution on molecules
Form when electrons are unequally shared
–Example: water molecule
–Polar molecules have regions with partial electrical charges resulting from unequal sharing of electrons among atoms
Areas of different partial charges attract one another, forming hydrogen bonds
–Much weaker than ionic or covalent bonds
–Results from unequal charge distribution on molecules
Form when electrons are unequally shared
–Example: water molecule
–Polar molecules have regions with partial electrical charges resulting from unequal sharing of electrons among atoms
Areas of different partial charges attract one another, forming hydrogen bonds
Chemical reactions
–Involve the formation or breaking of chemical bonds
–There are three basic types of chemical reactions involved in physiology:
•Synthesis reaction
–combining of two or more substances to form a more complex substance
–Formation of new chemical bonds
–A + B → AB
•Decomposition reaction
–Breaking down of a substance into two or more simpler substances
–Breaking of chemical bonds
–AB → A + B
•Exchange reaction
–Decomposition of two substances and, in exchange, synthesis of two new compounds from them
–AB + CD → AD + CB
•Reversible reactions—occur in both directions
–Involve the formation or breaking of chemical bonds
–There are three basic types of chemical reactions involved in physiology:
•Synthesis reaction
–combining of two or more substances to form a more complex substance
–Formation of new chemical bonds
–A + B → AB
•Decomposition reaction
–Breaking down of a substance into two or more simpler substances
–Breaking of chemical bonds
–AB → A + B
•Exchange reaction
–Decomposition of two substances and, in exchange, synthesis of two new compounds from them
–AB + CD → AD + CB
•Reversible reactions—occur in both directions
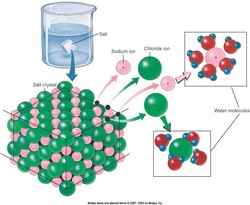
Inorganic compounds
—few have carbon atoms and none have C–C or C–H bonds
Organic molecules
–Have at least one carbon atom and at least one C–C or C–H bond in each molecule
–Often have functional groups attached to the carbon-containing core of the molecule
•Water
–The body’s most abundant and important compound
–Properties of water 1. High specific heat
•Water can lose and gain large amounts of heat with little change in its own temperature
•Enables the body to maintain a relatively constant temperature
2. High heat of vaporization
•Water requires absorption of significant amounts of heat to change water from a liquid to a gas
•Allows the body to dissipate excess heat
—few have carbon atoms and none have C–C or C–H bonds
Organic molecules
–Have at least one carbon atom and at least one C–C or C–H bond in each molecule
–Often have functional groups attached to the carbon-containing core of the molecule
•Water
–The body’s most abundant and important compound
–Properties of water 1. High specific heat
•Water can lose and gain large amounts of heat with little change in its own temperature
•Enables the body to maintain a relatively constant temperature
2. High heat of vaporization
•Water requires absorption of significant amounts of heat to change water from a liquid to a gas
•Allows the body to dissipate excess heat
•Electrolytes
–Large group of inorganic compounds, which includes acids, bases, and salts
–Substances that dissociate in solution to form ions
–Positively charged ions are cations
–Negatively charged ions are anions
Acids and bases
•Acids
–Any substance that releases a hydrogen ion (H+) when in solution; “proton donor”
–Level of “acidity” depends on the number of hydrogen ions a particular acid will release
•Bases
–Electrolytes that dissociate to yield hydroxide ions (OH–)
or other electrolytes that combine with hydrogen ions (H+)
–Described as “proton acceptors”
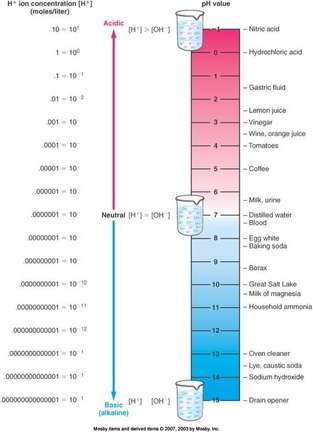
pH scale—measuring acidity and alkalinity
–pH indicates the degree of acidity or alkalinity of a solution
–pH of 7 indicates neutrality (equal amounts of H+ and OH–)
–pH of less than 7 indicates acidity
–pH of more than 7 indicates alkalinity
•Buffers
–Maintain the constancy of the pH
–Minimize changes in the concentrations of H+
and OH– ions
–Act as a “reservoir” for hydrogen ions
–Large group of inorganic compounds, which includes acids, bases, and salts
–Substances that dissociate in solution to form ions
–Positively charged ions are cations
–Negatively charged ions are anions
Acids and bases
•Acids
–Any substance that releases a hydrogen ion (H+) when in solution; “proton donor”
–Level of “acidity” depends on the number of hydrogen ions a particular acid will release
•Bases
–Electrolytes that dissociate to yield hydroxide ions (OH–)
or other electrolytes that combine with hydrogen ions (H+)
–Described as “proton acceptors”
pH scale—measuring acidity and alkalinity
–pH indicates the degree of acidity or alkalinity of a solution
–pH of 7 indicates neutrality (equal amounts of H+ and OH–)
–pH of less than 7 indicates acidity
–pH of more than 7 indicates alkalinity
•Buffers
–Maintain the constancy of the pH
–Minimize changes in the concentrations of H+
and OH– ions
–Act as a “reservoir” for hydrogen ions
“Organic” describes compounds that contain C–C or C–H bonds
•Carbohydrates
–Organic compounds containing carbon, hydrogen, and oxygen
–Commonly called sugars and starches
•Monosaccharides
–Simple sugars with short carbon chains; those with six carbons are hexoses (e.g., glucose), whereas those with five are pentoses (e.g., ribose, deoxyribose)
•Disaccharides and polysaccharides
–Two (di-) or more (poly-) simple sugars that are bonded together through a synthesis reaction
•Proteins
–Most abundant organic compounds
–Chainlike polymers
–Amino acids—building blocks of proteins
•Essential amino acids
–Eight amino acids that cannot be produced by the human body
•Nonessential amino acids
–12 amino acids can be produced from molecules available in the human body
•Lipids
–Water-insoluble organic molecules that are critically important biological compounds
–Major roles:
Energy source
Structural role
Integral parts of cell membranes
–Phospholipids
•Fat compounds similar to triglyceride
•One end of the phospholipid is water-soluble (hydrophilic); the other end is fat-soluble (hydrophobic)
•Phospholipids can join two different chemical environments
•Phospholipids may form double layers called bilayers that make up cell membranes
–Prostaglandins
•Commonly called “tissue hormones”; produced by cell membranes throughout the body
•Effects are many and varied; however, they are released in response to a specific stimulus and are then inactivated
•Nucleic Acids
–DNA (deoxyribonucleic acid)
•Composed of deoxyribonucleotides; that is, structural units composed of the pentose sugar (deoxyribose), phosphate group, and nitrogenous base (cytosine, thymine, guanine, or adenine)
•DNA molecule consists of two long chains of deoxyribonucleotides coiled into double-helix shape (Figure 2-27)
•Alternating deoxyribose and phosphate units form backbone of the chains
•Combined forms
–Examples:
•Adenosine triphosphate (ATP)—two extra phosphate groups to a nucleotide
•Lipoproteins—lipid and protein groups combined into a single molecule
•Glycoproteins—carbohydrate (glyco, “sweet”) and protein
•Carbohydrates
–Organic compounds containing carbon, hydrogen, and oxygen
–Commonly called sugars and starches
•Monosaccharides
–Simple sugars with short carbon chains; those with six carbons are hexoses (e.g., glucose), whereas those with five are pentoses (e.g., ribose, deoxyribose)
•Disaccharides and polysaccharides
–Two (di-) or more (poly-) simple sugars that are bonded together through a synthesis reaction
•Proteins
–Most abundant organic compounds
–Chainlike polymers
–Amino acids—building blocks of proteins
•Essential amino acids
–Eight amino acids that cannot be produced by the human body
•Nonessential amino acids
–12 amino acids can be produced from molecules available in the human body
•Lipids
–Water-insoluble organic molecules that are critically important biological compounds
–Major roles:
Energy source
Structural role
Integral parts of cell membranes
–Phospholipids
•Fat compounds similar to triglyceride
•One end of the phospholipid is water-soluble (hydrophilic); the other end is fat-soluble (hydrophobic)
•Phospholipids can join two different chemical environments
•Phospholipids may form double layers called bilayers that make up cell membranes
–Prostaglandins
•Commonly called “tissue hormones”; produced by cell membranes throughout the body
•Effects are many and varied; however, they are released in response to a specific stimulus and are then inactivated
•Nucleic Acids
–DNA (deoxyribonucleic acid)
•Composed of deoxyribonucleotides; that is, structural units composed of the pentose sugar (deoxyribose), phosphate group, and nitrogenous base (cytosine, thymine, guanine, or adenine)
•DNA molecule consists of two long chains of deoxyribonucleotides coiled into double-helix shape (Figure 2-27)
•Alternating deoxyribose and phosphate units form backbone of the chains
•Combined forms
–Examples:
•Adenosine triphosphate (ATP)—two extra phosphate groups to a nucleotide
•Lipoproteins—lipid and protein groups combined into a single molecule
•Glycoproteins—carbohydrate (glyco, “sweet”) and protein
CREDITS TO OWNER : http://moodle.cesa10.k12.wi.us/course/view.php?id=514

